Building Block Of Nucleic Acids
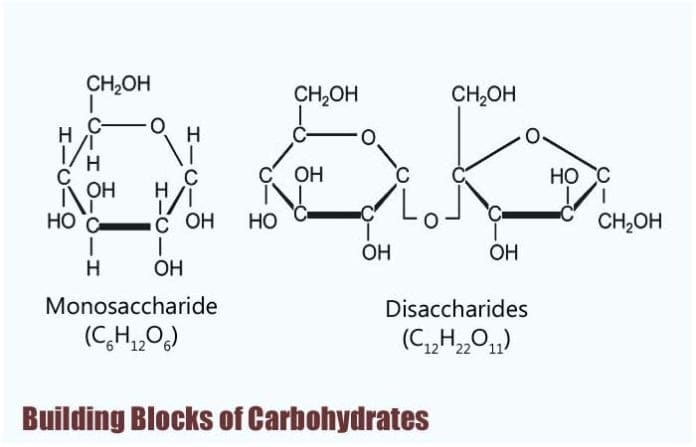
Building Blocks of Carbohydrates: The bones biochemistry of living organisms can, therefore, be understood regarding the morphology and physiology of the four biological macromolecules: carbohydrates, proteins, lipids, and nucleic acids.
Amid these iv macromolecules, carbohydrates are considered to be the most abundant as they serve as the immediate sources of energy of living organisms.
The discussion "carbohydrate" comes from the two Greek words "carbo" and "hydro" which hateful "carbon" or "coal" and "water" respectively. This probably came to the fact that when sugars are heated, carbon and h2o are released. In biochemical terms, they are referred to equally either polyhydroxy aldehydes or polyhydroxy ketones.
But what exactly brand upwards these complex macromolecules tha e them to acquit out such functions? In this commodity, allow'south explore about these carbohydrates and their biological building blocks: the monosaccharides.
Tabular array of Contents
- Building Blocks of Carbohydrates
- Physical and Chemical Properties of Monosaccharides
- Three Virtually Mutual Monosaccharides
- 1. Glucose
- ii. Fructose
- three. Galactose
- The Glycosidic Bond
- Polysaccharides
- one. Glycogen
- 2. Starch
- Function of Carbohydrates
- References
Building Blocks of Carbohydrates
Physical and Chemic Backdrop of Monosaccharides
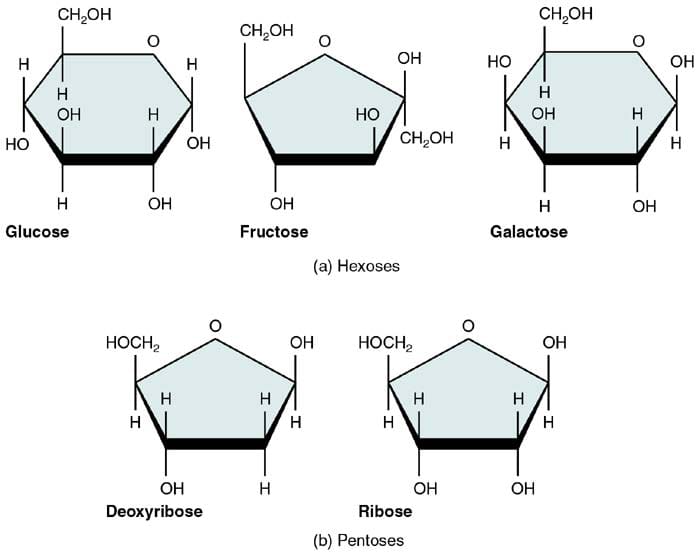
Monosaccharides are known to be the simplest form of carbohydrates, and as such, they are considered to be their edifice blocks.
The term "monosaccharide" comes from the Greek discussion "mono" which means "one" and "sugar" which means "sugar" or "sweetness."
This is because monosaccharides contain only one unit of polyhydroxy aldehyde or ketone and are grouped according to the number of carbon they have.
- In full general, monosaccharides, share the same chemical formula of Chalf dozenH12O6, and because having six carbon atoms, they are too called equally hexose.
- Existence sugars, monosaccharides naturally have a sweet taste (fructose is considered to be the sweetest amid them) and remain in their solid forms at room temperature.
- In spite of their very high molecular weights, they are very soluble in h2o as compared with other substances with the same molecular weight. The fact that at that place are a lot of OH groups in their structure makes this possible.
- Regarding their chemical limerick, monosaccharides do non usually exhibit their open up-chain structures. In this type of formation, an alcohol grouping can be readily added to the carbonyl grouping to create a pyranose ring that contains a stable conformation of a circadian hemiacetal or hemiketal.
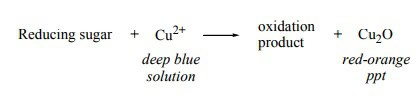
Monosaccharides, in general, tin exist readily oxidized by certain chemicals. The aldehydes and ketones in their structures contain OH groups located on the carbon adjacent to the carbonyl group that tin can react with the cupric ions (Cu) of Benedict's reagent. After this reaction, a formation of an orange precipitate of copper (I) oxide or Cu2O will occur.
All monosaccharides undergo this blazon of reaction and are called reducing sugars. (The chemic reaction is shown in a higher place)
![]()
Three Most Mutual Monosaccharides
In that location are iii about common naturally occurring monosaccharides: glucose, fructose, and galactose. Despite sharing the same chemical formula, they have different structural configurations, thus making them unlike regarding overall structure and part.
1. Glucose

Considered as the most critical monosaccharide, glucose is besides known as dextrose or claret saccharide. As it is, it serves as an immediate energy source during cellular respiration (photosynthesis).
- Glucose is naturally occurring in plants and animals in its free course. It is synthesized in a process called gluconeogenesis from non-carbohydrate molecules like glycerol and pyruvate. At the aforementioned time, it can also come from the break-down of glycogen in the procedure called glycogenolysis.
![]()
ii. Fructose

Also known as fruit sugar, fructose is the natural sugar that is found in fruits and honey. Overall, information technology is considered to be the sweetest among sugars. In chemical terms, fructose is besides called as levulose.
- Care should exist taken when consuming too much fructose as it is often associated with gastrointestinal issues and with somehow contributes to the increment in the fat content of claret.
![]()
iii. Galactose

Last but not the least is the monosaccharide galactose that is derived from the hydrolysis of the disaccharide lactose (milk-sugar). This lactose derived from milk is an essential energy source for many animals, including humans.
- Interestingly, the mammalian torso can convert glucose to galactose for the mammary glands to produce the lactose in milk.
![]()
The Glycosidic Bond
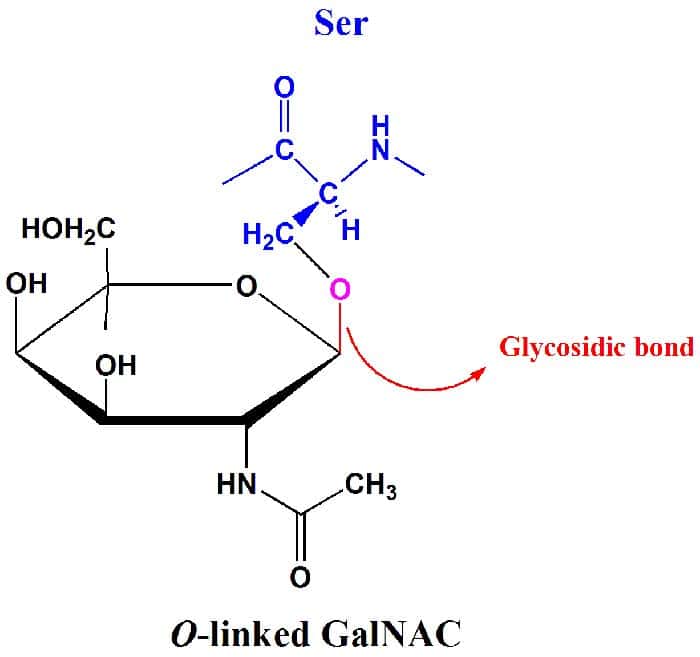
Because monosaccharides (and other carbohydrates) have many OH groups, they can be joined to one another by covalent bonds. In particular, glycosidic bonds are the type of covalent bonds that join together carbohydrate molecules with other groups, which may or may not be of the aforementioned type.
- As their name suggests, they are involved with glycosides which are band-shaped sugar molecules that may either be a five-membered band or a six-membered one.
- It should also be important to note that not all glycosidic bonds are the aforementioned: they can be either linked to a nitrogen or oxygen.
And just like what was alluded to higher up, the fact that monosaccharides contain many OH groups means that many linkages via glycosidic bonds are possible. Hence, the diversity of these ties can exist correlated with the vast array of monosaccharides, and their forms make more complex carbohydrates be packed with much information.
![]()
Polysaccharides

Long chains of polymeric saccharides that are formedvia the glycosidic linkages of monosaccharides are known as polysaccharides. Being complex sugars, they play essential roles in the maintenance of an organism's structural integrity as well every bit the storage of energy.
- Polysaccharides that are made up of the same type of monosaccharides are referred to every bit homopolymers.
- There are 2 almost common types of polysaccharides in animal and found cells: glycogen and starch. They are described beneath.
![]()
i. Glycogen

In animals, the well-nigh common type of homopolymer is glycogen. Glycogen is a very big polysaccharide made of glucose monomers and is considered to exist the storage form of carbohydrates in creature cells.
- Its glucose units are mostly linked via α-one,4-glycosidic bonds; however, at about every ten units, linkages via α-ane,6-glycosidic bonds are used.
![]()
two. Starch

On the other manus, starch is the storage form of energy in plants. It comes in 2 forms: amylose and amylopectin. These two types are readily hydrolyzed by the enzyme (called α-amylase) produced by the salivary glands in the rima oris and by the pancreas.
- Amylose is the unbranched type and is composed of glucose units linked via α-1,iv glycosidic bonds. Contrary, amylopectin is the branched type and is connected via α-1,six glycosidic bonds, in the same mode, that glycogen is formed.
However, you don't always need many monosaccharide units to form a polysaccharide. A special type of polysaccharide, called a disaccharide, is fabricated up of just two monosaccharide units linked via a glycosidic bail. The most common disaccharides are sucrose (glucose and fructose), lactose (glucose and galactose), and maltose (two glucose).
![]()
Function of Carbohydrates
As alluded to earlier, carbohydrates serve every bit the immediate energy sources. In college organisms, they serve to enable the metabolism of fats to avert the breaking down of proteins for free energy. Aside from that carbohydrates are likewise needed to metabolize fats. Plainly, if the body generates enough free energy for its physiological functions, actress energy becomes stored as fat.
![]()
In conclusion, monosaccharide as sugar building blocks can be linked to a wide diversity of stereochemistries which are essential in the formation of more complex structures. Because of these small units, living organisms are supplied with ample corporeality of energy that helps them survive. Indeed, small things brand a large difference.
![]()
Cite This Page
References
- "Sugar definition and meaning | Collins English Dictionary". Accessed November 21, 2017. Link.
- "Complex Carbohydrates Are Formed by Linkage of Monosaccharides – Biochemistry – NCBI Bookshelf". Accessed November 21, 2017. Link.
- "Food Tests – Bridegroom'southward Examination for Reducing Carbohydrate – Bright Biology Pupil". Accessed Nov 21, 2017. Link.
- "Glycogenolysis and glycogenesis – Metabolism, insulin and other hormones – Diapedia, The Living Textbook of Diabetes". Accessed November 21, 2017. Link.
- "Fructose is generated in the human encephalon | YaleNews". Accessed November 21, 2017. Link.
- "Open Learning Initiative: Register for a Class". Accessed Nov 21, 2017. Link.
- "Glycogen Biosynthesis; Glycogen Breakdown". Accessed Nov 21, 2017. Link.
- "PDB-101: Alpha-amylase". Accessed November 21, 2017. Link.
Building Block Of Nucleic Acids,
Source: https://www.bioexplorer.net/building-blocks-of-carbohydrates.html/
Posted by: moffettpenated.blogspot.com


0 Response to "Building Block Of Nucleic Acids"
Post a Comment